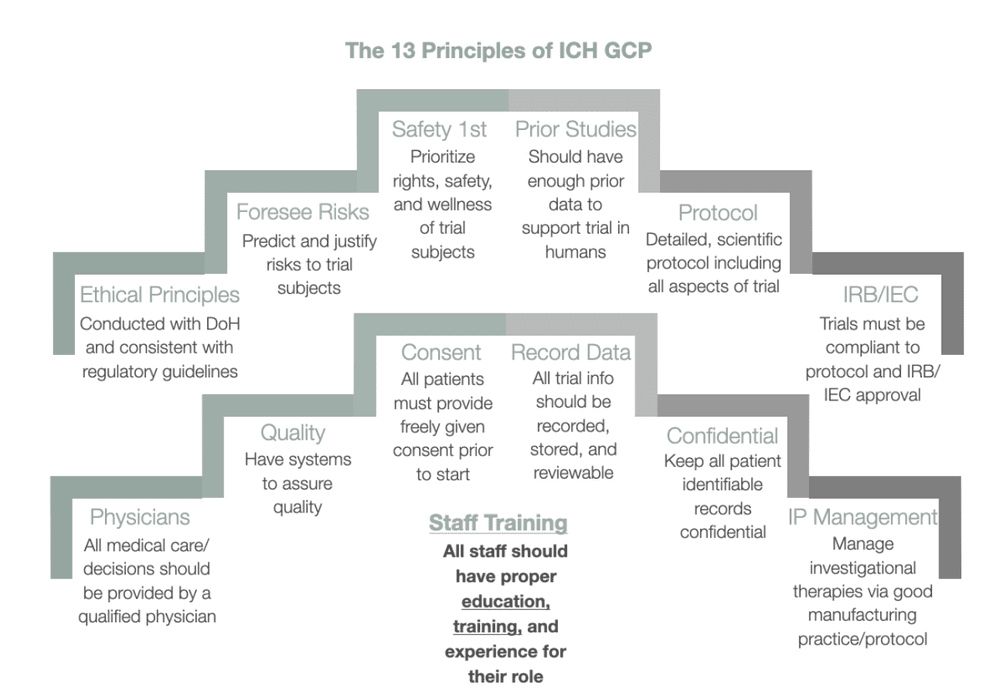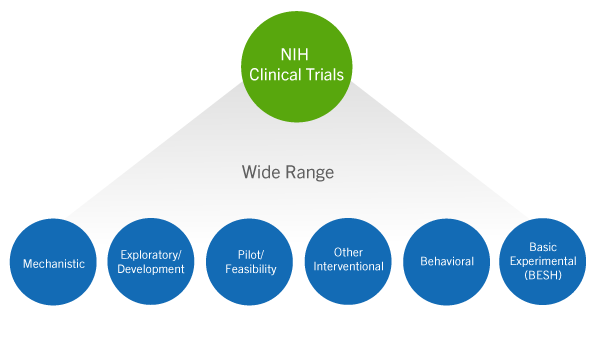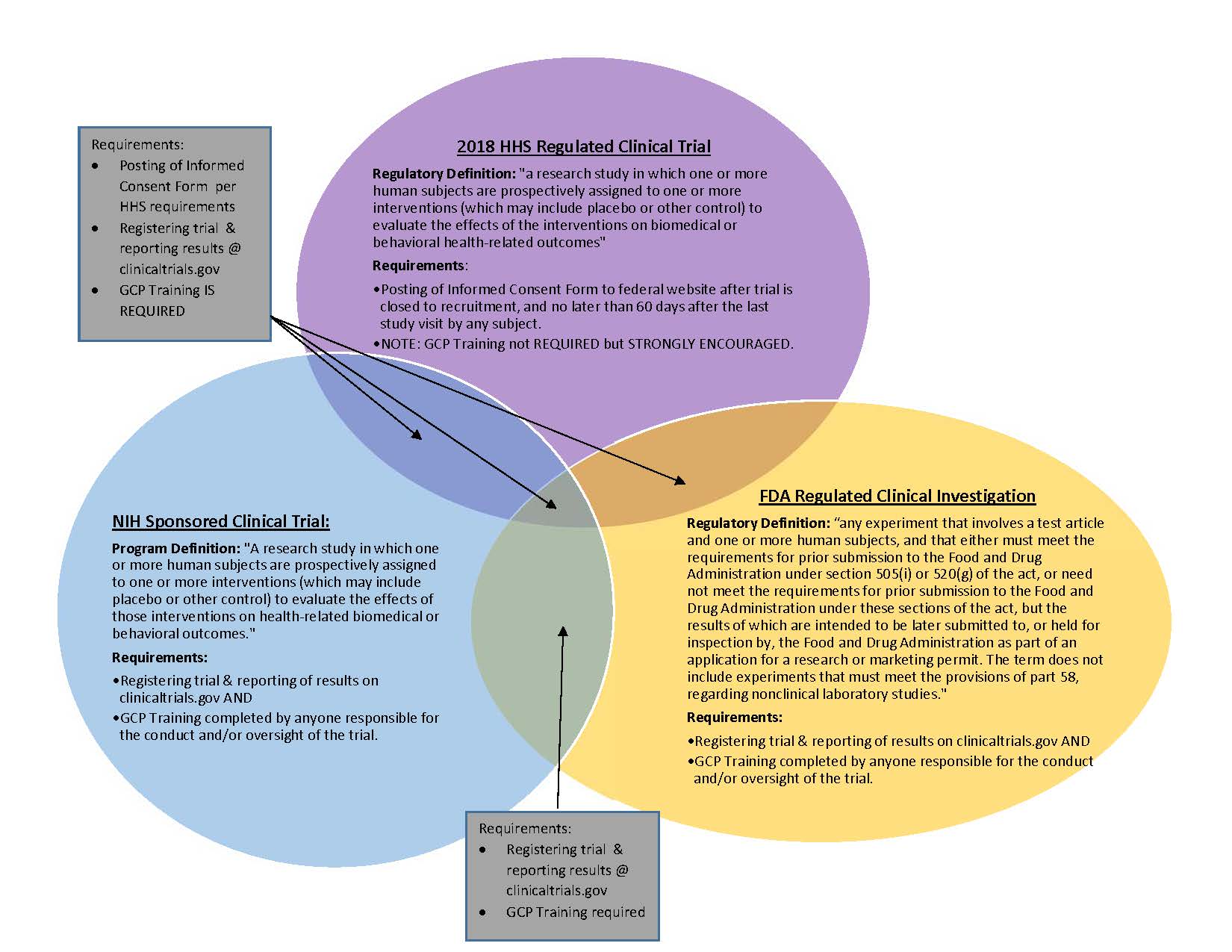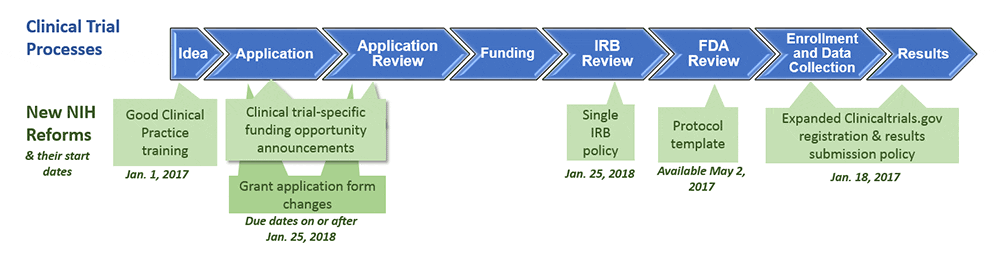
NIH Redefines Clinical Trials and Sets New Requirements: Is Your Human Subjects Research Affected? - ASHG
Issues, challenges, and the way forward in conducting clinical trials among neonates: investigators' perspective | Journal of Perinatology