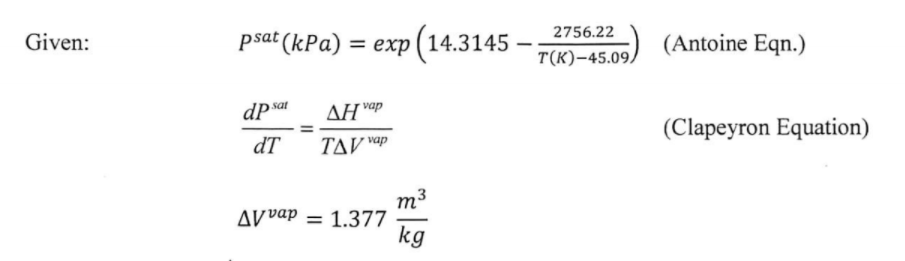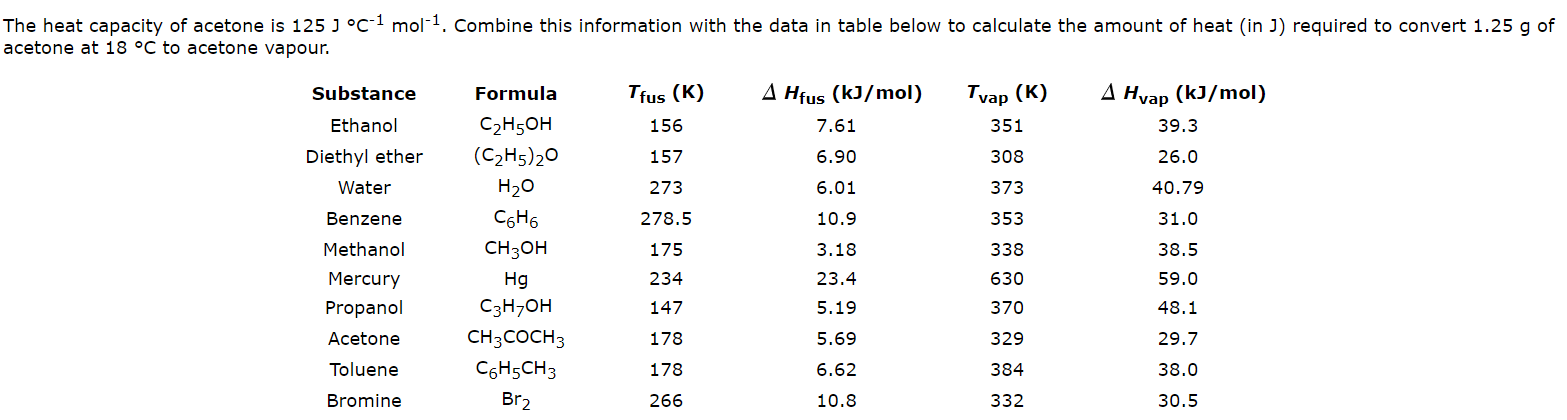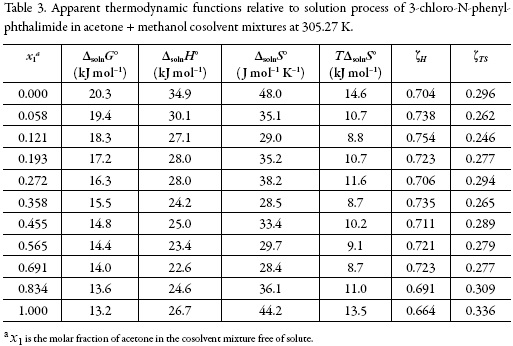
Solution thermodynamics and preferential solvation of 3-chloro-N-phenyl-phthalimide in acetone + methanol mixtures

Heat of vaporization of acetone. Simulation data: • this work, AUA4... | Download Scientific Diagram

A theoretical analysis on enthalpy of vaporization: Temperature-dependence and singularity at the critical state - ScienceDirect

OneClass: 2) The normal boiling point of acetone is 56.5°C At an elevation of 5300 ft, the atmospher...

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram
How to calculate the vapor pressure of acetone at 25.0°C if the enthalpy of vaporization for acetone is 32.0 kJ/mol and the normal boiling point of acetone is 56.5°C - Quora

Heat of vaporization of acetone. Simulation data: • this work, AUA4... | Download Scientific Diagram
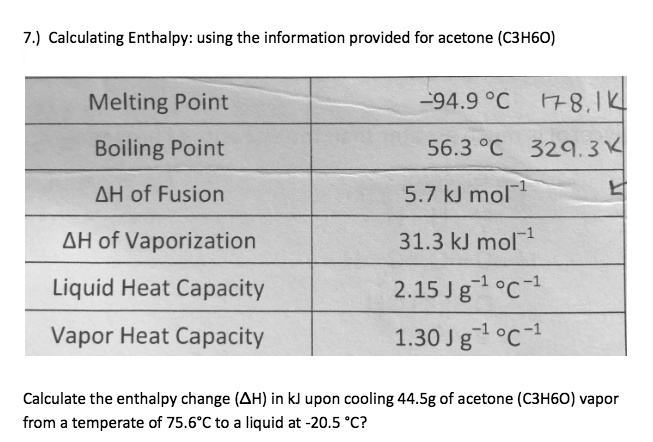
SOLVED:7.) Calculating Enthalpy: using the information provided for acetone (C3HGO) Melting Point Boiling Point AH of Fusion ~94.9 %€ +7-8,1444 56.3 "C 329.34 5.7 kJ mol-1 AH of Vaporization 31.3 kJ mol-1

`Delta_(vap)`S1 of acetone is `93.0 JK^(-1) "mol"^(-1)`.If boiling point of acetone is `56^(@)C`, calculate the heat required for the vaporisation of 1 g of acetone.




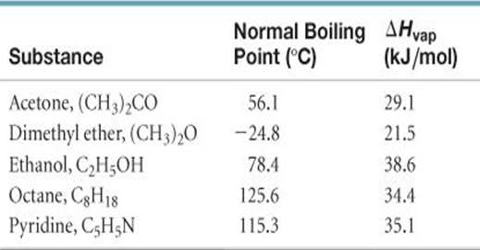
![Solved] Calculate ΔĤ for each of the following processes. In each case, res | SolutionInn Solved] Calculate ΔĤ for each of the following processes. In each case, res | SolutionInn](https://s3.amazonaws.com/si.question.images/images/question_images/1590/0/5/6/4075ec655d7a186e1590056399297.jpg)